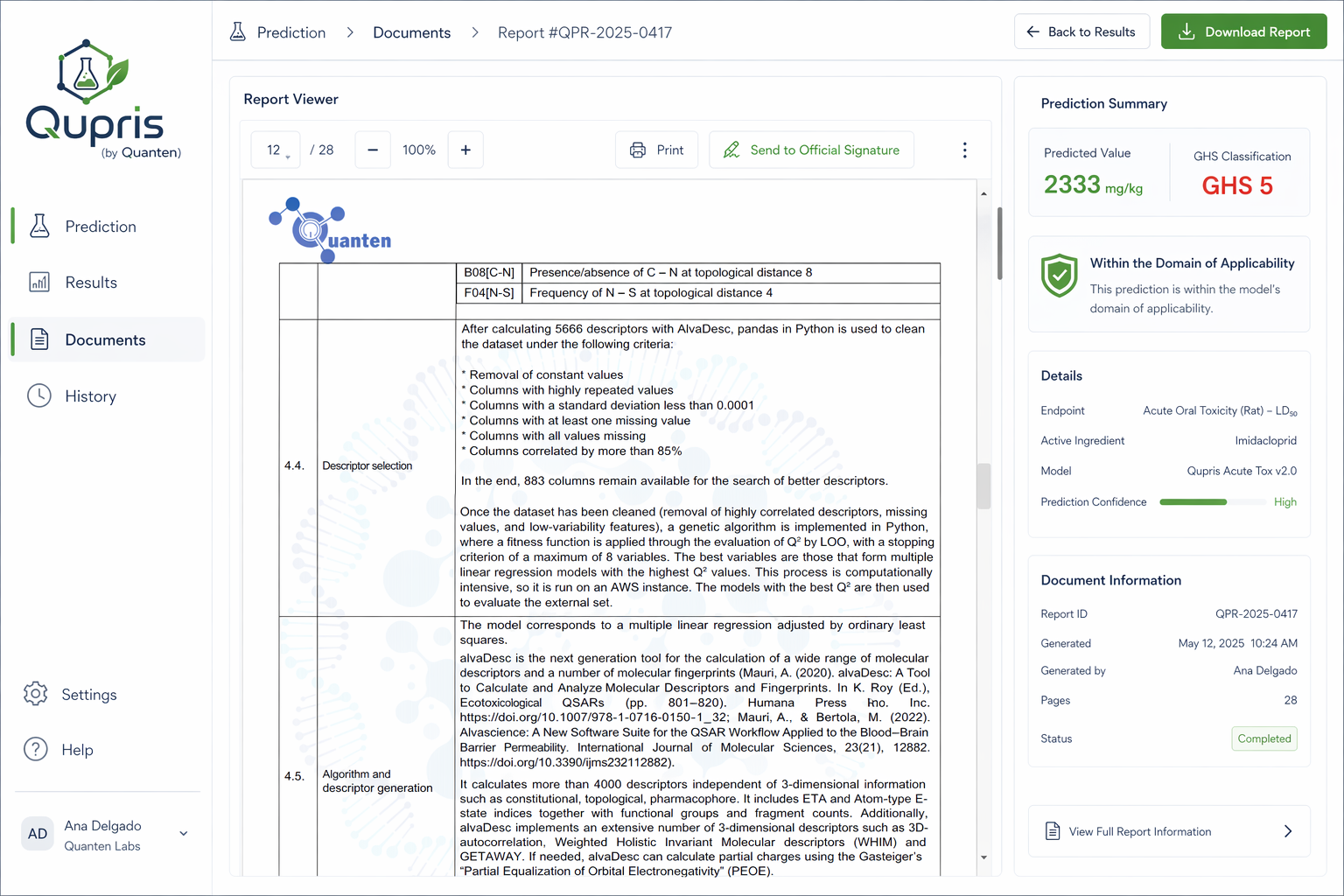
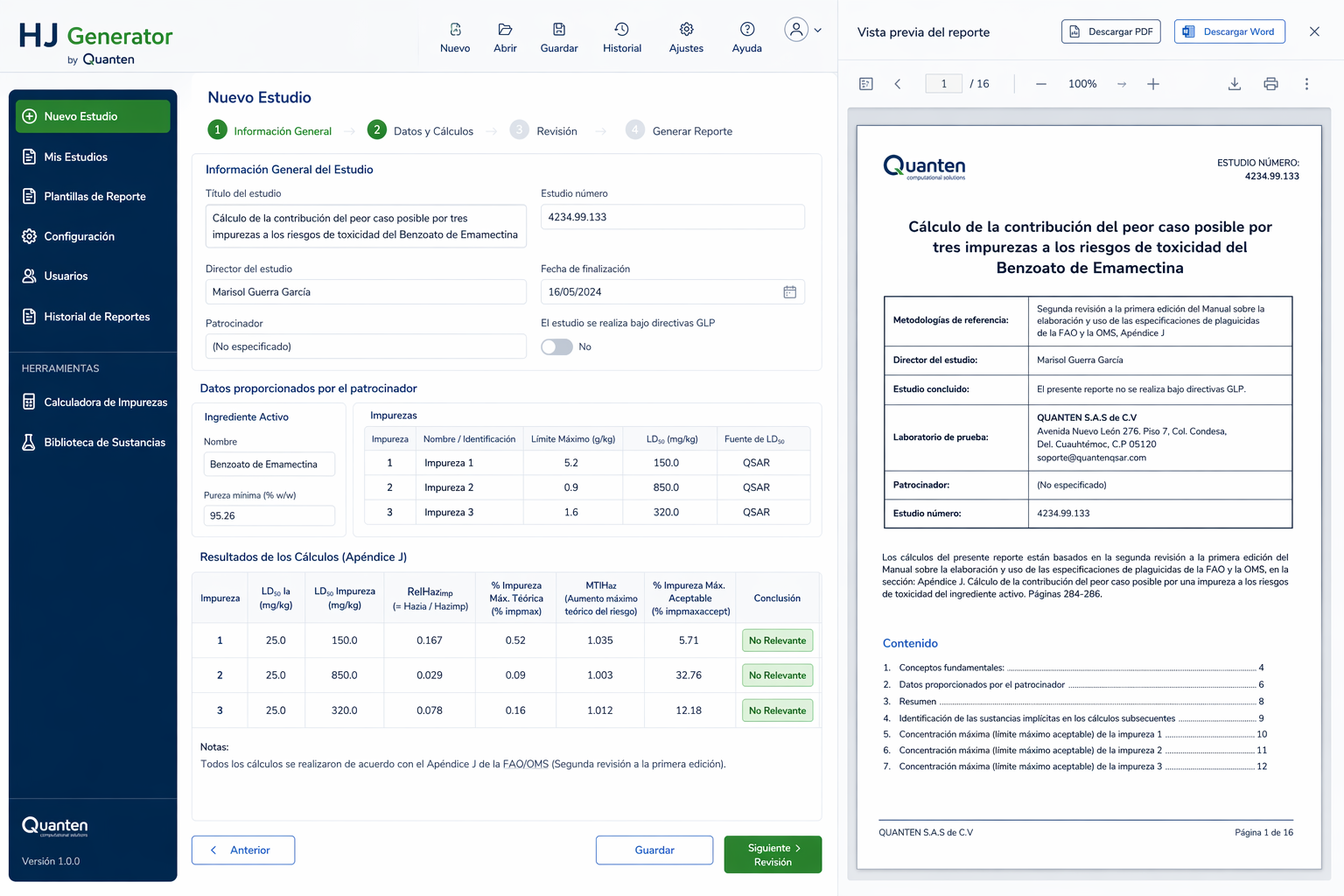
QSAR and QSPR Modeling
The continuous expansion of the chemical industry has led to a significant increase in the manufacture, importation, and use of chemical substances worldwide. While this growth supports innovation and industrial development, the introduction of new substances without adequate evaluation may pose risks to human health and the environment.
To address these concerns, regulatory frameworks have been established to ensure proper hazard identification and safe chemical management. One of the most important is Regulation (EC) No 1907/2006, commonly known as REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals). This legislation requires companies to register substances and submit technical documentation covering areas such as intended uses, classification and labeling, safe handling recommendations, and toxicological or ecotoxicological information.
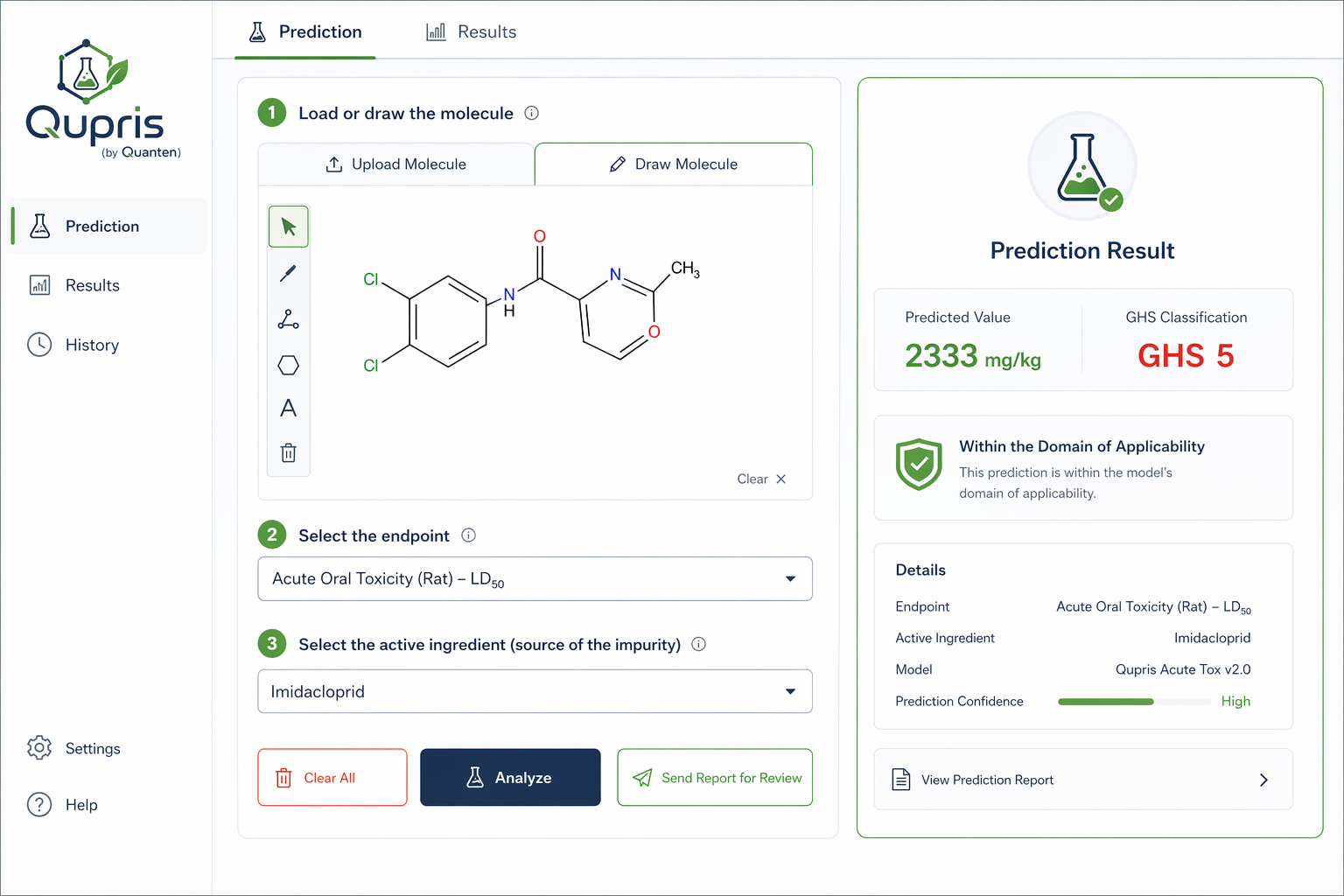
Although hazard data can be generated through conventional laboratory studies, REACH strongly encourages the use of alternative assessment strategies whenever scientifically justified. Among these, computational methods such as QSAR modeling and related in silico approaches—offered by Quanten QSAR—provide efficient solutions for estimating a broad range of endpoints while reducing costs, shortening timelines, and minimizing the need for animal testing.
ENDPOINTS
Physico-chemical properties
| REACH | Endpoint | TEST |
|---|
| 7.2 | Melting/freezing point | OECD 102 |
| 7.3 | Boiling point | OECD 103 |
| 7.4 | Relative density | OECD 109 |
| 7.5 | Vapour pressure | OECD 104 |
| 7.6 | Surface tension | OECD 115 |
| 7.7 | Water solubility | OECD 105 |
| 7.8 | Partition coefficient n-octanol/water | OECD 107, 117, 123 |
| 7.9 | Flash-point | EU A.9 |
| 7.10 | Flammability | EU A.10, EU A.12, EU A.13 |
| 7.11 | Explosive properties | EU A.14 |
| 7.12 | Self-ignition temperature | EU A.15 |
| 7.13 | Oxidising properties | EU A.17 |
| 7.14 | Granulometry | |
| 7.15 | Stability in organic solvents and identity of relevant degradation products | |
| 7.16 | Dissociation constant | OECD 112 |
| 7.17 | Viscosity | OECD 114 |
Toxicity
| REACH | Endpoint | TEST |
|---|
| 8.1 | Skin irritation or skin corrosion | OECD 439, 430, 431, 435, EU B.40, EU B.40 bis |
| 8.1.1 | In vivo skin irritation | OECD 439, 430, 431, 435 |
| 8.2 | Eye irritation | EU B.47, OECD 437, EU B.48, OECD 438, OECD 460, OECD 491, OECD 492 |
| 8.2.1 | In vivo eye irritation | OECD 405 |
| 8.3 | Skin sensitisation | OECD 429, 406, 442A, 442B, 442C, 442D, 442E, |
| 8.4 | Mutagenicity | OECD 473 |
| 8.4.1 | In vitro gene mutation study in bacteria | OECD 471 |
| 8.4.2 | In vitro cytogenicity study in mammalian cells or in vitro micronucleus study | OECD 487 |
| 8.4.3 | In vitro gene mutation study in mammalian cells | OECD 490 |
| 8.5 | Acute toxicity | |
| 8.5.1 | By oral route | OECD 420, 423, 425 |
| 8.5.2 | Acute toxicity By inhalation | OECD 403, 433, 436 |
| 8.5.3 | Acute toxicity By dermal route | OECD 402 |
| 8.6 | Repeated dose toxicity | |
| 8.6.1 | Short-term repeated dose toxicity study (28 days), one species, male and female, most appropriate route of administration, having regard to the likely route of human exposure. | OECD 422 |
| 8.6.2 | Sub-chronic toxicity study (90- day), one species, rodent, male and female, most appropriate route of administration, having regard to the likely route of human exposure. | |
| 8.7 | Reproductive toxicity | |
| 8.7.1 | Screening for reproductive/developmental toxicity, one species | OECD 421, 422 |
| 8.7.2 | Pre-natal developmental toxicity study, one species, most appropriate route of administration, having regard to the likely route of human exposure | OECD 414 |
| 8.7.3 | Two-generation reproductive toxicity study, one species, male and female, most appropriate route of administration, having regard to the likely route of human exposure | OECD 416 |
| 8.9.1 | Carcinogenicity study | OECD 451, 453 |
Ecotoxicity
| REACH | Endpoint | TEST |
|---|
| 9.1 | Aquatic toxicity | |
| 9.1.1 | Short-term toxicity testing on invertebrates (preferred species Daphnia) | OECD 202 |
| 9.1.2 | Growth inhibition study aquatic plants (algae preferred) | OECD 201, 221 |
| 9.1.3 | Short-term toxicity testing on fish: the registrant may consider long-term toxicity testing instead of short- term. | OECD 203, OECD 236, OECD 210 |
| 9.1.4 | Activated sludge respiration inhibition testing | OECD 209 |
| 9.1.5 | Long-term toxicity testing on invertebrates (preferred species Daphnia) | OECD Guideline 211/ EU Method C.20 |
| 9.1.6 | Long-term toxicity testing on fish | |
| 9.1.6.1 | Fish early-life stage (FELS) toxicity test | OECD TG 210 |
| 9.1.6.2 | Fish short-term toxicity test on embryo and sac-fry stages | OECD 212 |
| 9.1.6.3 | Fish, juvenile growth test | OECD 215 |
| 9.2.1 | Degradation (biotic) | |
| 9.2.1.1 | Ready biodegradability | OECD 301 |
| 9.2.1.2 | Simulation testing on ultimate degradation in surface water | OECD 309 |
| 9.2.1.3 | Soil simulation testing | OECD 307 |
| 9.2.1.4 | Sediment simulation testing | OECD 308 |
| 9.2.2.1 | Hydrolysis as a function of pH | OECD 111 |
| 9.2.3 | Identification of degradation products | |
| 9.3.1 | Adsorption/desorption screening | OECD 106, OECD 121 |
| 9.3.2 | Bioaccumulation in aquatic species, preferably fish | OECD 305 |
| 9.4 | Effects on terrestrial organisms | |
| 9.4.1 | Short-term toxicity to invertebrates | OECD 214 |
| 9.4.2 | Effects on soil micro-organisms | |
| 9.4.3 | Short-term toxicity to plants | |
| 9.4.4 | Long-term toxicity testing on invertebrates | |
| 9.4.6 | Long-term toxicity testing on plants | |
| 9.5.1 | Long-term toxicity to sediment organisms | |
| 9.6.1 | Long-term or reproductive toxicity to birds | |
Digital Toxicity Assessment
At Quanten QSAR, we provide advanced computational toxicology solutions to support the registration, development, and regulatory evaluation of chemical substances, pharmaceuticals, and impurities. Our expertise combines predictive science with practical regulatory strategy to accelerate decision-making and reduce experimental burden.
We offer toxicological profiling through validated in silico methodologies, including QSAR modeling, read-across, expert review, and weight-of-evidence approaches for endpoints such as mutagenicity, acute toxicity, sensitization, and systemic hazards.
For pharmaceutical and chemical development projects, we perform theoretical impurity elucidation using tools such as Zeneth, enabling the prediction of likely degradation pathways, transformation products, and potential process-related impurities under stress or storage conditions.
We also support ICH M7 assessments for mutagenic impurities, including structure review, QSAR expert systems, TTC-based classification, risk justification, and preparation of regulator-ready documentation.
Our digital assessments help clients save time, lower costs, and generate scientifically robust evidence for regulatory submissions worldwide.
Predictive Modeling & AI
At Quanten QSAR, we integrate advanced data science, machine learning, and artificial intelligence to transform complex datasets into strategic insights for research, product development, and business growth. Our solutions are designed to improve decision-making through accurate, scalable, and scientifically grounded predictive models.
We develop custom regression, classification, and pattern-recognition models for applications such as toxicity prediction, property estimation, formulation optimization, trend detection, market intelligence, and process performance analysis. Techniques may include random forests, gradient boosting, neural networks, support vector machines, and interpretable statistical modeling.
Our team also applies AI-driven data mining and visualization to uncover hidden relationships, prioritize opportunities, and optimize R&D pipelines. Every project emphasizes transparency, validation, and practical business value.
Whether the objective is scientific innovation or operational efficiency, Quanten QSAR converts data into measurable advantage.
Specialized Training Programs
The scientific team at Quanten QSAR offers advanced training programs for professionals, universities, research groups, and companies seeking practical skills in computational sciences. Courses can be tailored by objectives and experience level, covering QSAR modeling, computational toxicology, chemoinformatics, machine learning, regulatory applications, impurity risk assessment, data analysis, and scientific software workflows.
Through case studies, hands-on exercises, and real-world applications, participants gain skills that can be immediately applied in research and industry.
Genomic Data Analysis
We provide advanced computational genomic solutions that convert complex biological datasets into clear, actionable knowledge for research, healthcare, biotechnology, and precision medicine initiatives. By combining bioinformatics, statistics, and artificial intelligence, we help organizations extract maximum value from genomic information.
Our services include sequence analysis, variant annotation and interpretation, biomarker discovery, gene expression studies, multi-omics data integration, and predictive modeling for clinically or commercially relevant outcomes. We also apply AI-driven methods to identify hidden patterns, prioritize targets, and accelerate scientific discovery.
Whether working with next-generation sequencing (NGS), population datasets, or translational research programs, we deliver reliable analyses that support innovation, product development, and evidence-based decisions.
From raw sequencing data to strategic insight, we turn genomic complexity into measurable opportunity.
R&D Scientific Consulting
We provide specialized scientific consulting to support research and development projects across the chemical, pharmaceutical, agrochemical, cosmetic, and biotechnology sectors. Our services combine deep scientific expertise with computational tools to accelerate decision-making, reduce uncertainty, and optimize innovation pathways.
From early-stage concept evaluation to advanced technical problem solving, we assist companies with formulation challenges, impurity strategies, predictive assessments, data interpretation, process optimization, and regulatory-oriented scientific guidance.
Whether you need an external expert perspective or long-term technical collaboration, Quanten QSAR delivers strategic, science-driven solutions for Mexico and the world.
Supervised Learning
We develop predictive models using labeled data to solve classification and regression problems across scientific and industrial applications. Our expertise includes toxicity prediction, property modeling, quality control, trend forecasting, and data-driven decision support using modern machine learning techniques.
Unsupervised Learning
We apply advanced data exploration techniques to uncover hidden patterns, relationships, and structures within complex datasets. Our services include clustering, dimensionality reduction, anomaly detection, segmentation, and knowledge discovery for scientific, industrial, and business applications.
Molecular Docking & Virtual Screening
We provide advanced computational strategies to identify and prioritize the most promising compounds before costly experimental testing. Through molecular docking and virtual screening, we evaluate large numbers of molecules to predict their potential interactions, affinity, and selectivity toward biological targets.
These approaches accelerate discovery programs by focusing resources on candidates with the highest probability of success for subsequent in vitro and in vivo evaluation. Our services support both structure-based drug design (SBDD), where target protein structures guide compound selection, and ligand-based drug design (LBDD), where known active molecules are used to identify new opportunities.
Applications include hit identification, lead optimization, repurposing strategies, binding mode analysis, and prioritization of chemical libraries for pharmaceutical, biotech, and research projects.
By integrating computational efficiency with scientific insight, we help reduce development time, lower screening costs, and improve decision-making in early-stage innovation.
Read-Across
Read-Across service card included as requested in the services grid, with icon reference readac.png.
Regulatory Affairs for Mexico and Latin America
Quanten QSAR provides specialized regulatory consulting for companies seeking market access, compliance, and product continuity in Mexico, Latin America, and global markets. We combine regulatory expertise with scientific depth to deliver efficient, evidence-based solutions.
Registration & Market Access
- New product registrations
- Renewals and extensions
- Variations, modifications, and updates
- Lifecycle management of existing authorizations
- Expansion into new markets across the region
Sectors We Support
- Pesticides and agrochemicals
- Plant nutrients and fertilizers
- Pharmaceuticals
- Medical devices
- Cosmetics and personal care
- Veterinary products
- Biocides and industrial chemicals
Regulatory Documentation
- Technical dossier preparation and review
- Data-gap analysis
- Classification strategies
- Labeling and compliance review
- Scientific responses to authority observations
Permits & Compliance
- Operating notices and sanitary licenses
- Import permits and related authorizations
- Advertising notifications
- Regulatory audits and readiness assessments
- Ongoing compliance support
Scientific Advantage
Unlike traditional consultants, Quanten QSAR strengthens submissions through:
- Computational toxicology
- QSAR modeling
- Impurity risk assessment
- Predictive data analysis
- Technical interpretation of complex studies
- Risk-based regulatory strategy
Authorities & Regional Support
Experience supporting processes before COFEPRIS, SEMARNAT, SADER, SENASICA, and comparable agencies throughout Latin America.
Why Quanten QSAR
We help clients reduce delays, prevent costly mistakes, anticipate regulatory objections, and accelerate approvals through science-driven consulting.